Not all high-fitness sequences have plausible evolutionary paths from lower-fitness starting points via sequential introduction of mutations
I first saw this precise idea articulated by Weinreich et al. in their aptly named "Darwinian evolution can follow only very few mutational paths to fitter proteins"[1]. Many mutation combinations don't have additive effects on every aspect governing protein fitness (expression, stability, function, etc.), and Weinreich et al. noted that introducing a subset of mutations from a high-fitness variant onto a lower-fitness starting sequence could reduce fitness rather than raise it:
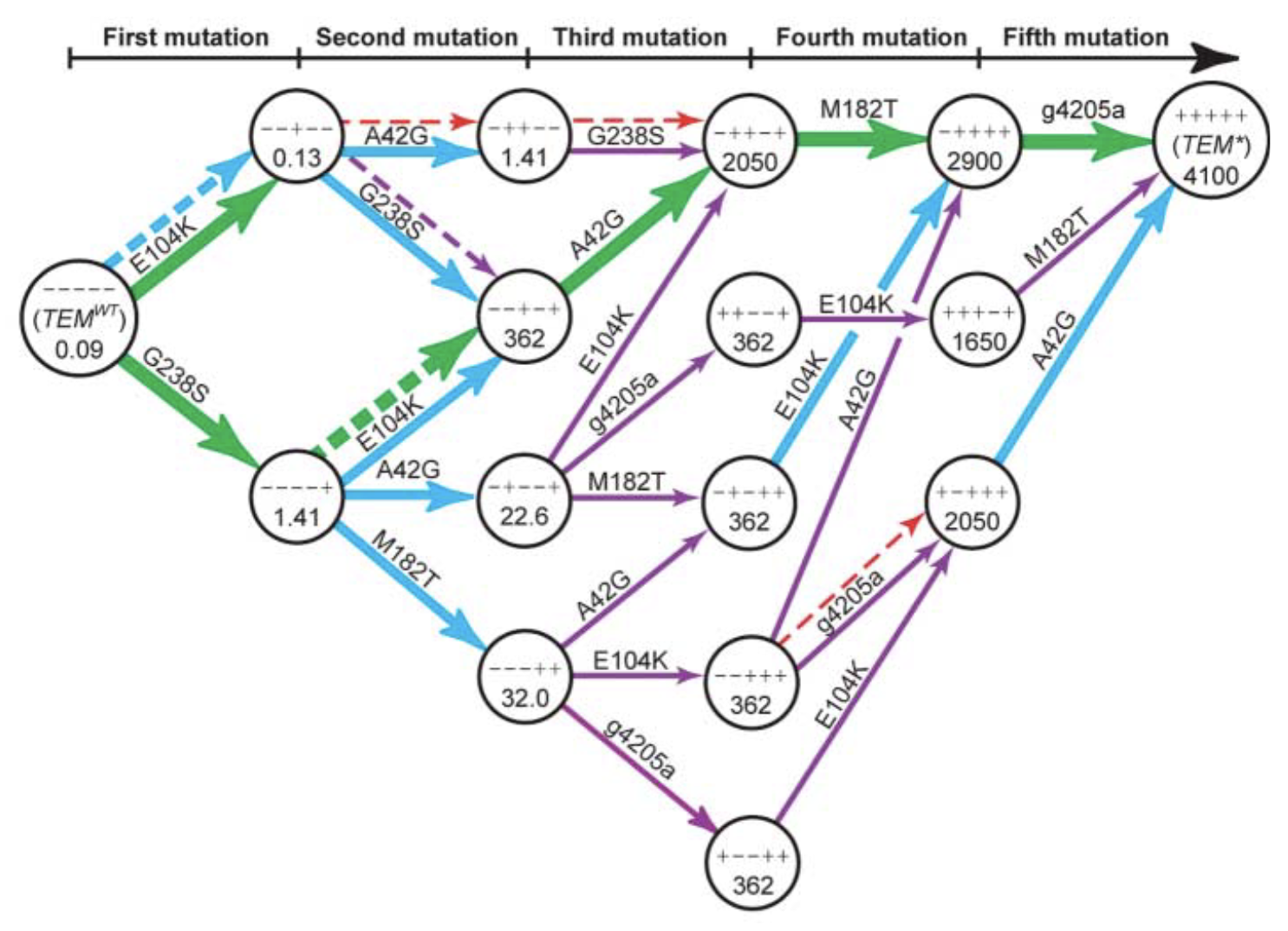
Here's another version of this figure published nearly twenty years later by Frances Arnold's lab[2]:
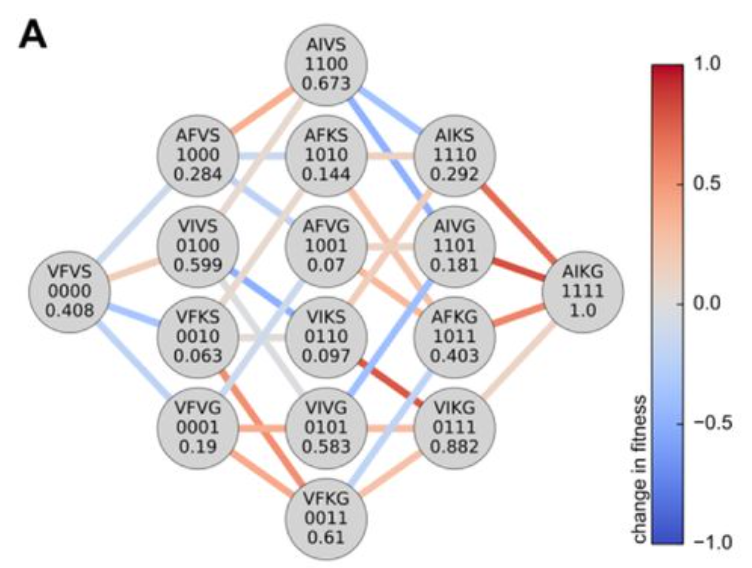
The FuncLib paper[3], introduces one interesting case study. The authors identified four mutations in a phosphotriesterase that drastically increased catalytic activity. Yet none of the sequences with any incomplete combination of these four mutations were as fit as either the starting or final sequence.
Note that this isn't necessarily the case for all proteins, or of every specific property of any given protein (for example antibodies).
References
Weinreich, D. M., Delaney, N. F., DePristo, M. A., & Hartl, D. L. (2006). Darwinian Evolution Can Follow Only Very Few Mutational Paths to Fitter Proteins. Science, 312(5770), 111–114. https://doi.org/10.1126/science.1123539 ↩︎
Johnston, K. E., Almhjell, P. J., Watkins-Dulaney, E. J., Liu, G., Porter, N. J., Yang, J., & Arnold, F. H. (2024). A combinatorially complete epistatic fitness landscape in an enzyme active site. Proceedings of the National Academy of Sciences, 121(32). https://doi.org/10.1073/pnas.2400439121 ↩︎
Khersonsky, O., Lipsh, R., Avizemer, Z., Ashani, Y., Goldsmith, M., Leader, H., Dym, O., Rogotner, S., Trudeau, D. L., Prilusky, J., Amengual-Rigo, P., Guallar, V., Tawfik, D. S., & Fleishman, S. J. (2018). Automated Design of Efficient and Functionally Diverse Enzyme Repertoires. Molecular Cell, 72(1), 178-186.e5. https://doi.org/10.1016/j.molcel.2018.08.033 ↩︎